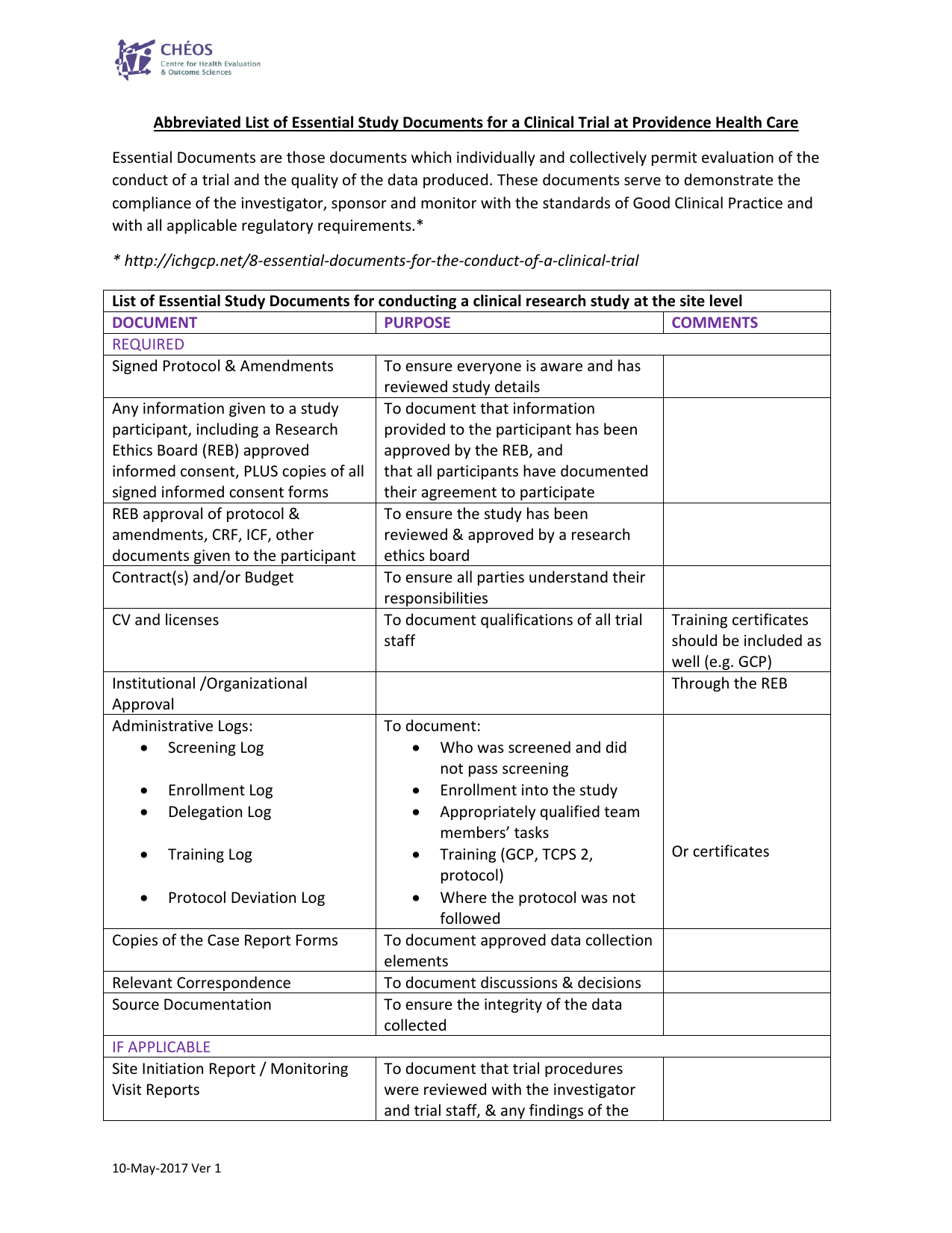
Medical writing for regulatory submission in clinical research and its challenges pdf2 by Medical Writing Experts - Issuu
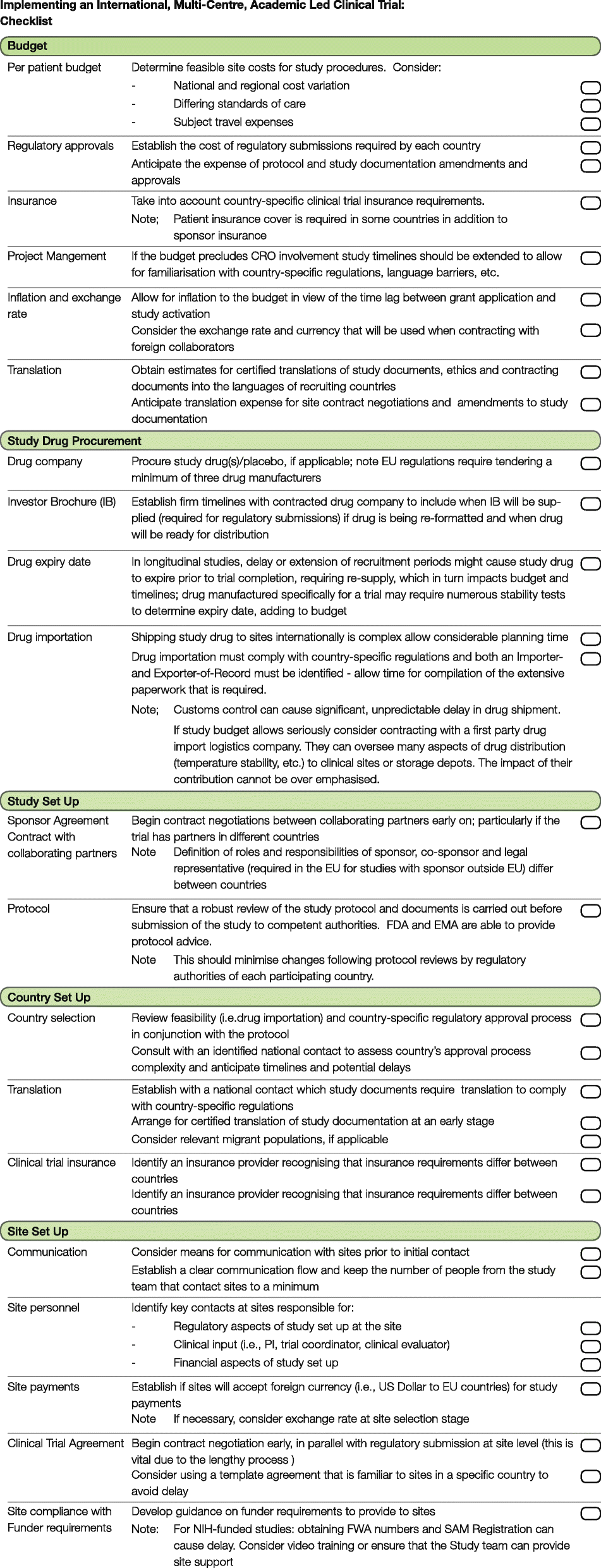
A checklist for clinical trials in rare disease: obstacles and anticipatory actions—lessons learned from the FOR-DMD trial | Trials | Full Text
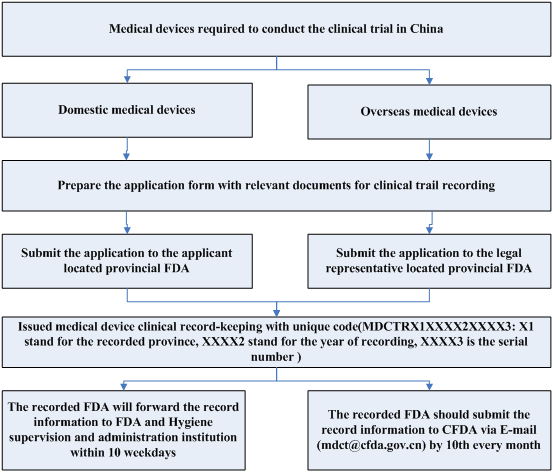
Clinical trial activities required to be recorded in China local FDA - Regulatory News - Medical Devices - CIRS Group
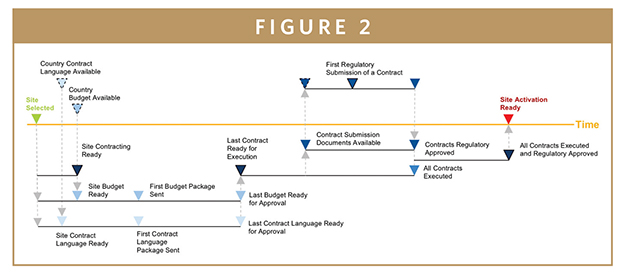
CLINICAL TRIALS - Spotlight on Quality in Study Startup: Automated Workflows Encourage Upfront Planning & Downstream Improvements in the eTMF

Clinical trials were missing from regulatory documents of extended-release methylphenidate for ADHD in adults: a case study of public documents - Journal of Clinical Epidemiology

What you Need and When – The Key Documents in the Drug Lifecycle - Trilogy Writing & Consulting GmbH
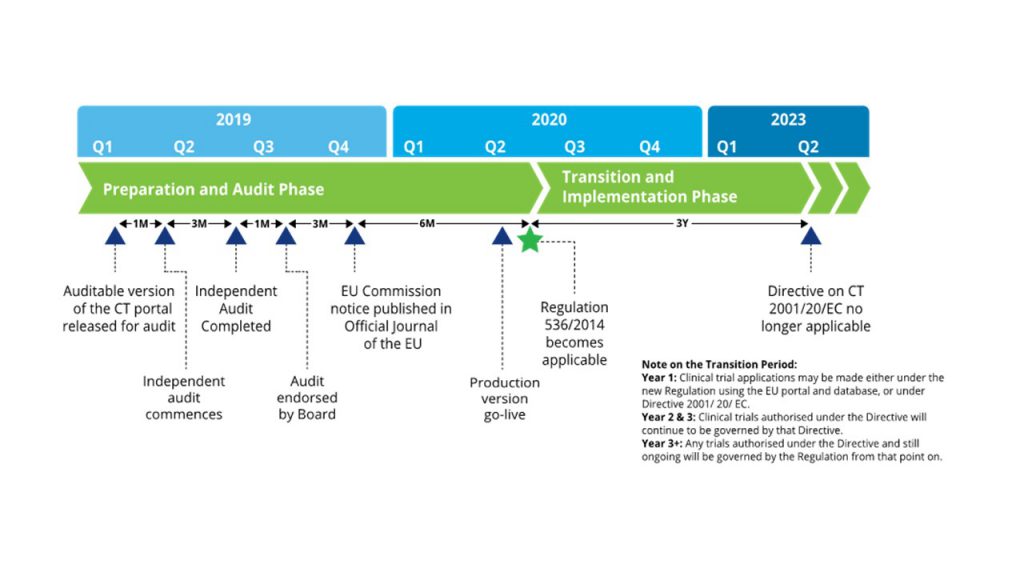
ADVANCED CLINICAL TRIALS THE CLINICAL TRIAL PROCESS: IMPENDING CHANGES IN THE REGULATORY FRAMEWORK - ADVANCED CLINICAL TRIALS

REGULATORY “ESSENTIAL” DOCUMENTATION Role of the RESEARCH COORDINATOR Best Practices 21CFR Part 11 Monday, November 7, ppt download