Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data | PLOS Medicine
PLOS Medicine: Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data

Clinical comparison between trial participants and potentially eligible patients using electronic health record data: A generalizability assessment method - ScienceDirect

A Comparison of Incremental Costs of Breast Cancer Clinical Trials to Standard of Care | Semantic Scholar
![PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/62bcc7885dac29517026f3ba577045d3b93dbb7b/2-Table2-1.png)
PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar
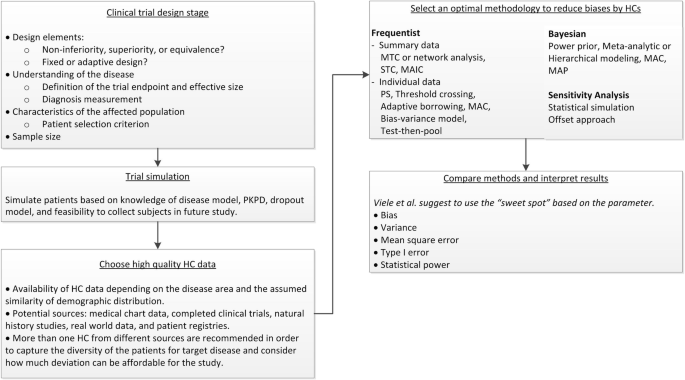
A roadmap to using historical controls in clinical trials – by Drug Information Association Adaptive Design Scientific Working Group (DIA-ADSWG) | Orphanet Journal of Rare Diseases | Full Text

Comparison of phase II and phase III clinical trial results. Displayed... | Download Scientific Diagram
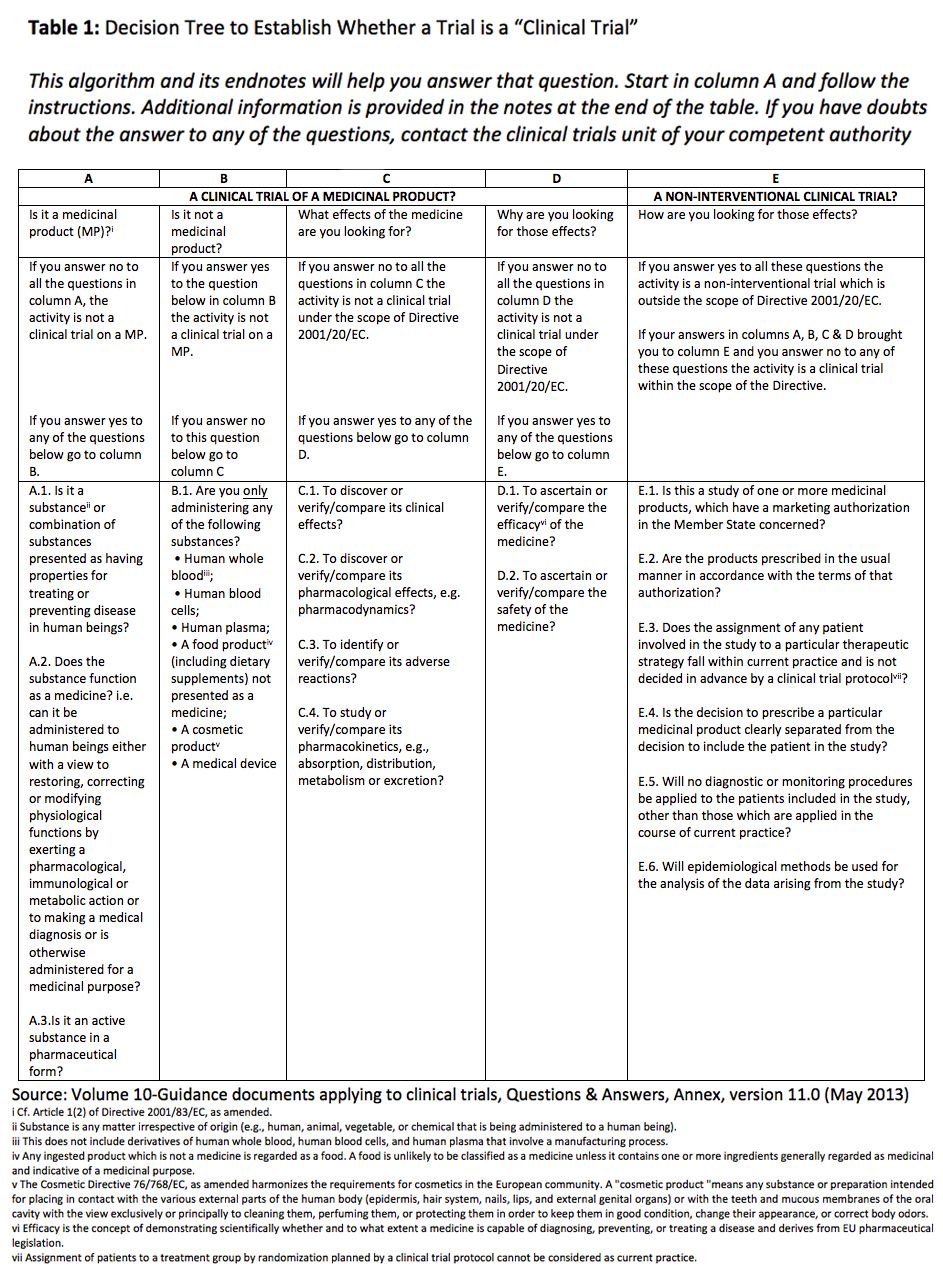
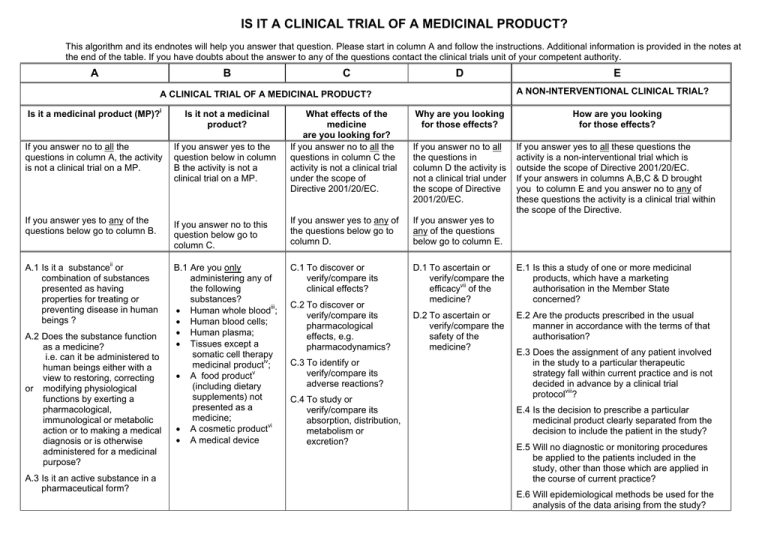
Interventional vs. Non-interventional Study Classification in the EU: Considerations on the Impact of Direct-to-Patient Contacts

60% cheaper clinical trials in Australia compared to US * CILIQUE - Clinical Outsourcing + Contract Management + CRO Vendor Selection & Management

Comparison of a randomized controlled trial, pragmatic clinical trial,... | Download Scientific Diagram

Quality Management of Electronic Systems in Clinical Trial Investigations: A Comparison of FDA and EMA Guidance - Life Science Training Institute

Clinical Trial Network - Did You Know? Clinical trials are research studies that test how well new medical approaches work in people. Each study answers scientific questions and tries to find better