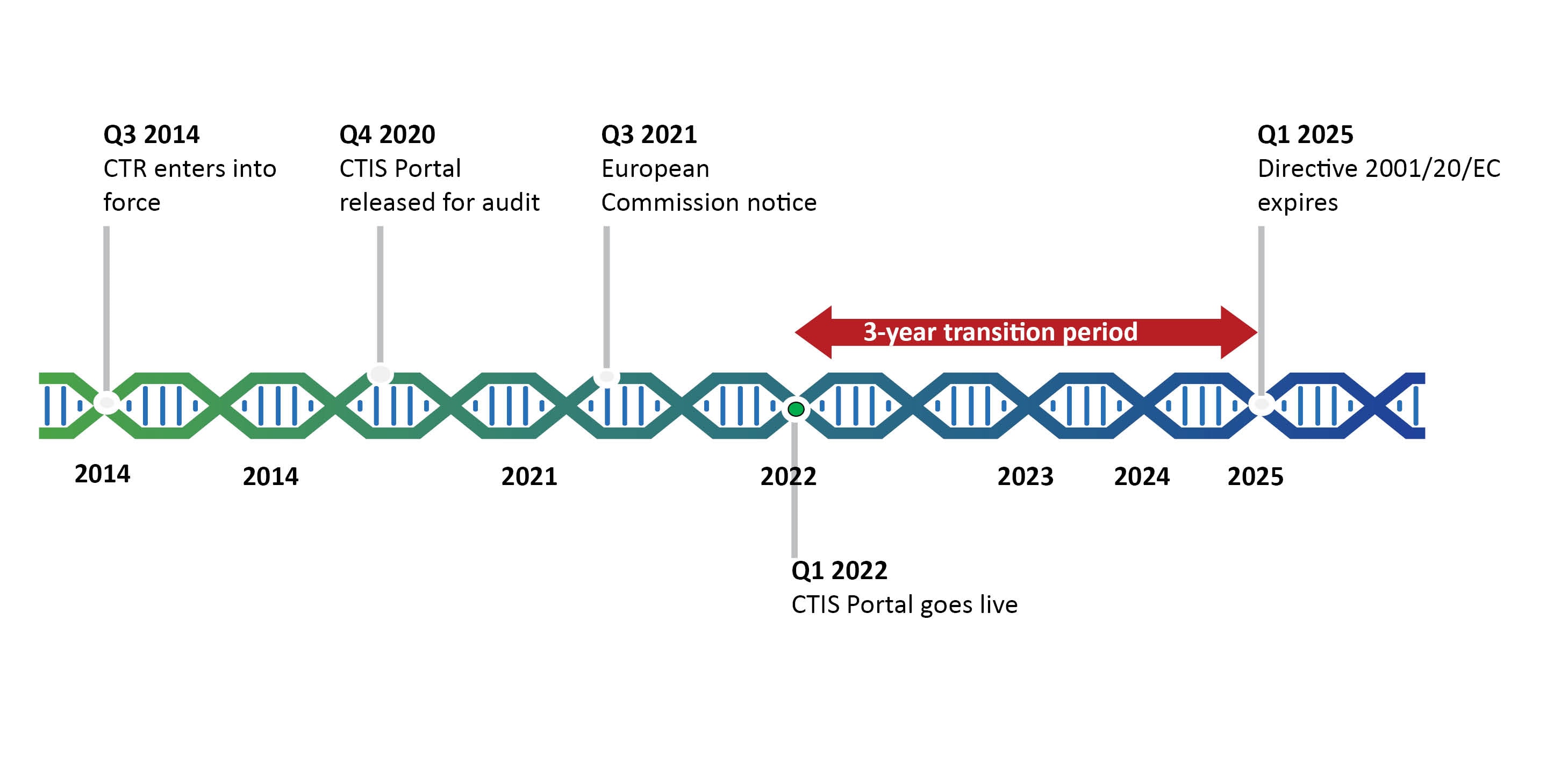
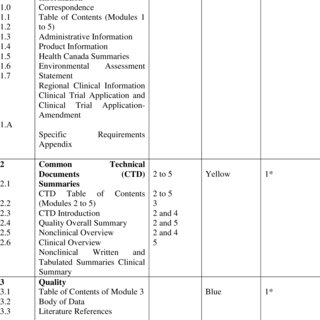
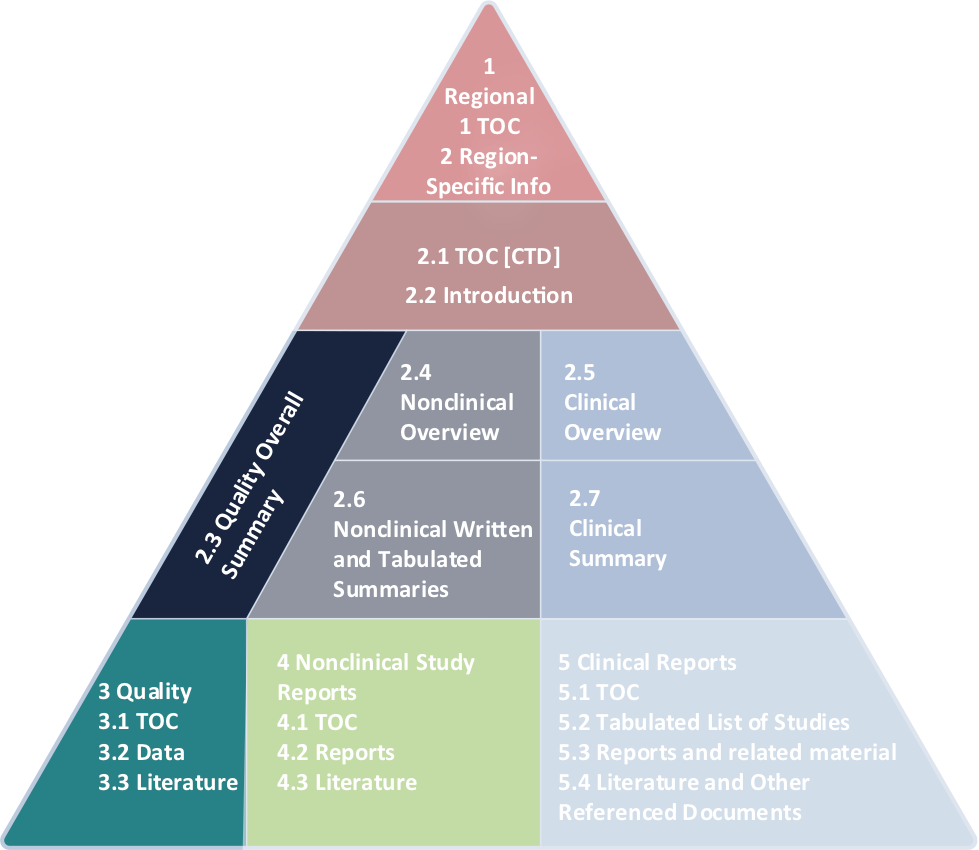
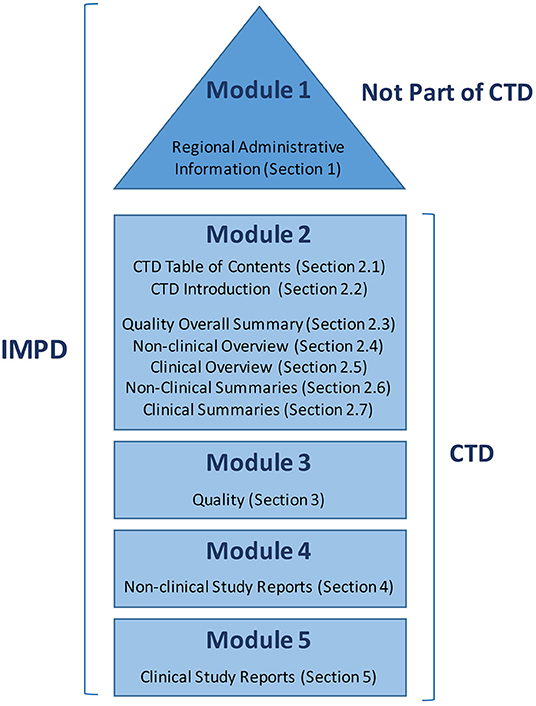
ClinProve - CTD Structure The Common Technical Document is divided into five modules: 1. Administrative and prescribing information 2. Overview and summary of modules 3 to 5 3. Quality (pharmaceutical documentation) 4.

The Challenge of CTD Submissions and Responding to Questions from the Authorities - Trilogy Writing & Consulting GmbH

POS-426 CLINICAL TRIAL DATABASE (CTD): INTEGRATED DATABASE MANAGEMENT SYSTEM FOR CLINICAL TRIALS - Kidney International Reports

What you Need and When – The Key Documents in the Drug Lifecycle - Trilogy Writing & Consulting GmbH

Preparation of a Preclinical Dossier to Support an Investigational New Drug (IND) Application and First-In-Human Clinical Trial - ScienceDirect

SUB04: Preparing Submissions in the Common Technical Document (CTD) Format | Zenosis – Learning for Life
Frontiers | Transitioning From Preclinical Evidence to Advanced Therapy Medicinal Product: A Spanish Experience | Cardiovascular Medicine

Representation of the components of the CTD. The nonclinical components... | Download Scientific Diagram
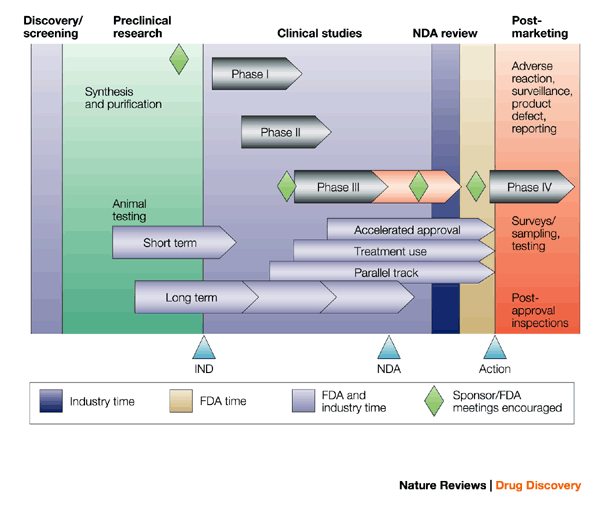
The Common Technical Document: the changing face of the New Drug Application | Nature Reviews Drug Discovery

A STUDY OF PROCEDURES FOR DOSSIER PREPARATION AND THEIR MARKETING AUTHORISATION IN DIFFERENT COUNTRIES OF SELECTED DRUG(S) | PharmaTutor