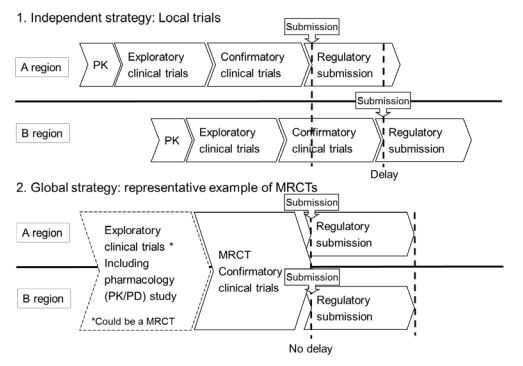
Strategic inclusion of regions in multiregional clinical trials - Seung Yeon Song, Deborah Chee, EunYoung Kim, 2019
E17 General Principles for Planning and Design of Multiregional Clinical Trials Guidance for Industry

Clinical Data Interchange Standards Consortium (CDISC) and the Multi-Regional Clinical Trials Center of Brigham and Women's Hospital (MRCT Center) Collaborate to Offer Plain Language Clinical Research Definitions as a New Standard

Advancing International Pediatric Clinical Research Webinar Series - The Multi-Regional Clinical Trials Center of Brigham and Women's Hospital and Harvard
E17 General Principles for Planning and Design of Multiregional Clinical Trials Guidance for Industry

The necessary conduct: Exploratory multiregional clinical trials in East Asia - Jeon - 2021 - Clinical and Translational Science - Wiley Online Library

Amazon.co.jp: Multiregional Clinical Trials for Simultaneous Global New Drug Development (Chapman & Hall/CRC Biostatistics Series) : Chen, Joshua, Quan, Hui: Foreign Language Books

Multi-Regional Clinical Trials Center of Brigham and Women's Hospital and Harvard (MRCT Center) | LinkedIn

Amazon.co.jp: Multiregional Clinical Trials for Simultaneous Global New Drug Development (Chapman & Hall/CRC Biostatistics Series) : Chen, Joshua, Quan, Hui: Foreign Language Books

FDA's treatment of China-developed drugs spurs demands for multiregional clinical trials | pharmaphorum
Regulatory and statistical issues of Multi- regional Clinical Trials: "Reference Cases" and current situation in Japan

HanchorBio Announces US IND Clearance for the Multi-Regional Clinical Trial of HCB101 to Treat Solid and Hematological Malignancies | BioSpace